DEPT NMR or APT NMR: Which is Better?
Distortionless Enhancement by Polarization Transfer NMR ( DEPT NMR) and Attached Proton Test NMR (APT NMR) are 13C NMR experiments that reveal the number of protons that are attached to individual 13C carbon atoms in a molecule.
DEPT NMR Advantages
DEPT is a polarization-transfer experiment in which the relaxation is governed by the T1 of the protons instead of the carbons. That is notable since the T1 relaxation time of protons is generally much shorter than that of the carbon 13 nuclei. So you may be able to save time with a DEPT NMR experiment.
There are several DEPT variants – DEPT-45, DEPT-90 and DEPT-135 NMR. Running two or more DEPT experiments can help distinguish between CH and CH3 more effectively than the APT (more on this later).
DEPT NMR Spectra Explained
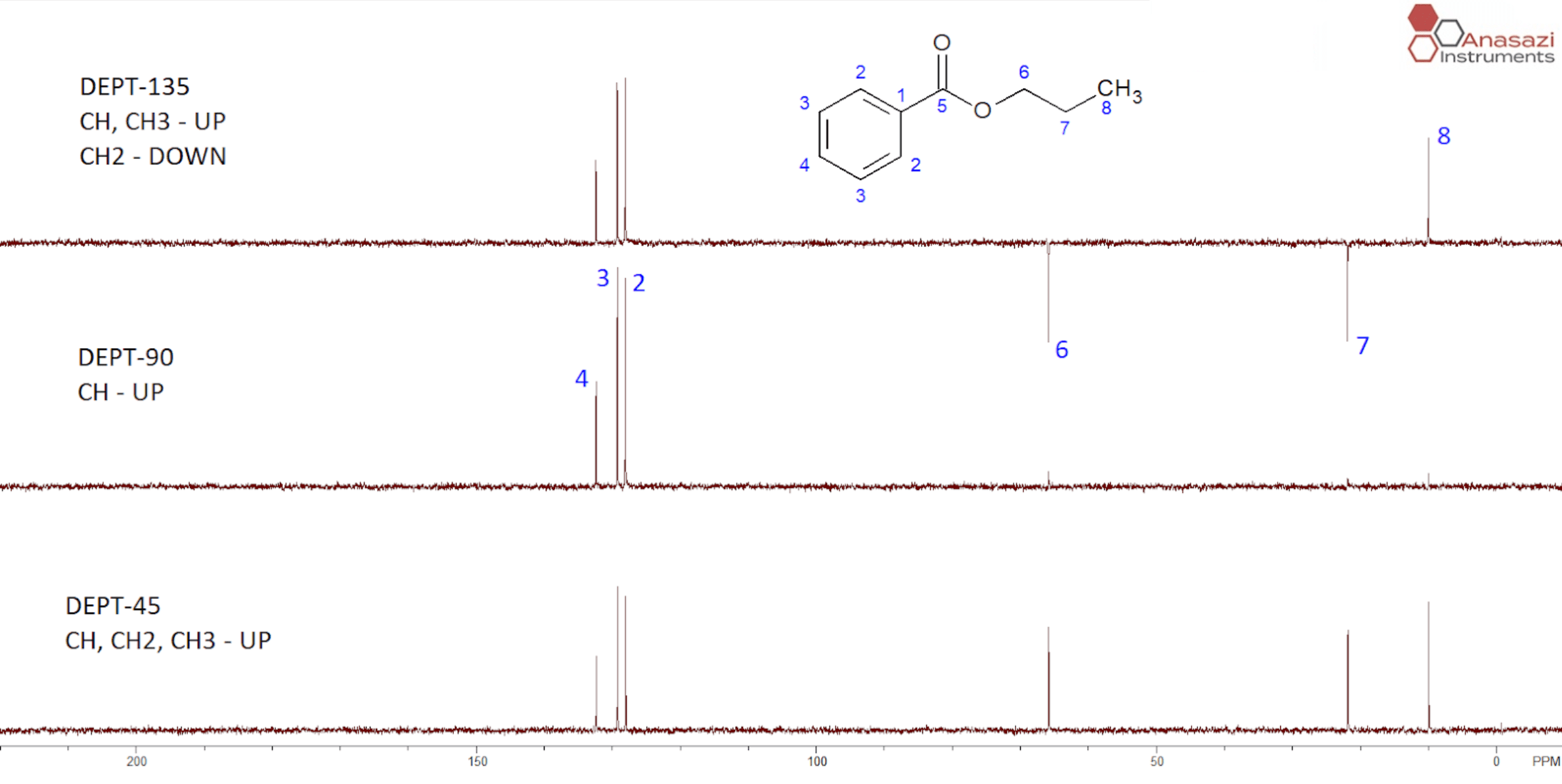
Figure 1 shows three spectra from a standard DEPT sequence. On some modern NMR spectrometers, the three spectra (DEPT-45, DEPT-90 and DEPT-135) are acquired and auto-processed with a single command (DEPT).
1. Only CH peaks show up in the DEPT-90.
– Identify the CH peaks in the middle spectrum below (2, 3 and 4).
– Identify any peaks pointing up in the DEPT-135 that were not present in the DEPT-90. These will be CH3s (8).
– Identify any inverted peaks in the DEPT-135 (6 and 7).
Figure 1
DEPT NMR Disadvantages
Using DEPT-135, DEPT-90 and DEPT-45 we have identified the primary, secondary and tertiary carbons. But where are the quaternary carbons (1) and (5)? Their peaks are absent in any of the DEPT spectra. This is the main disadvantage of the DEPT sequences – we cannot observe carbon nuclei that do not have directly attached protons.
As we saw earlier, you may be able to save time with DEPT. However, it also takes time to run the different variants of the DEPT to distinguish multiplicity. So depending on your needs it might actually not save any time.
APT NMR Advantages
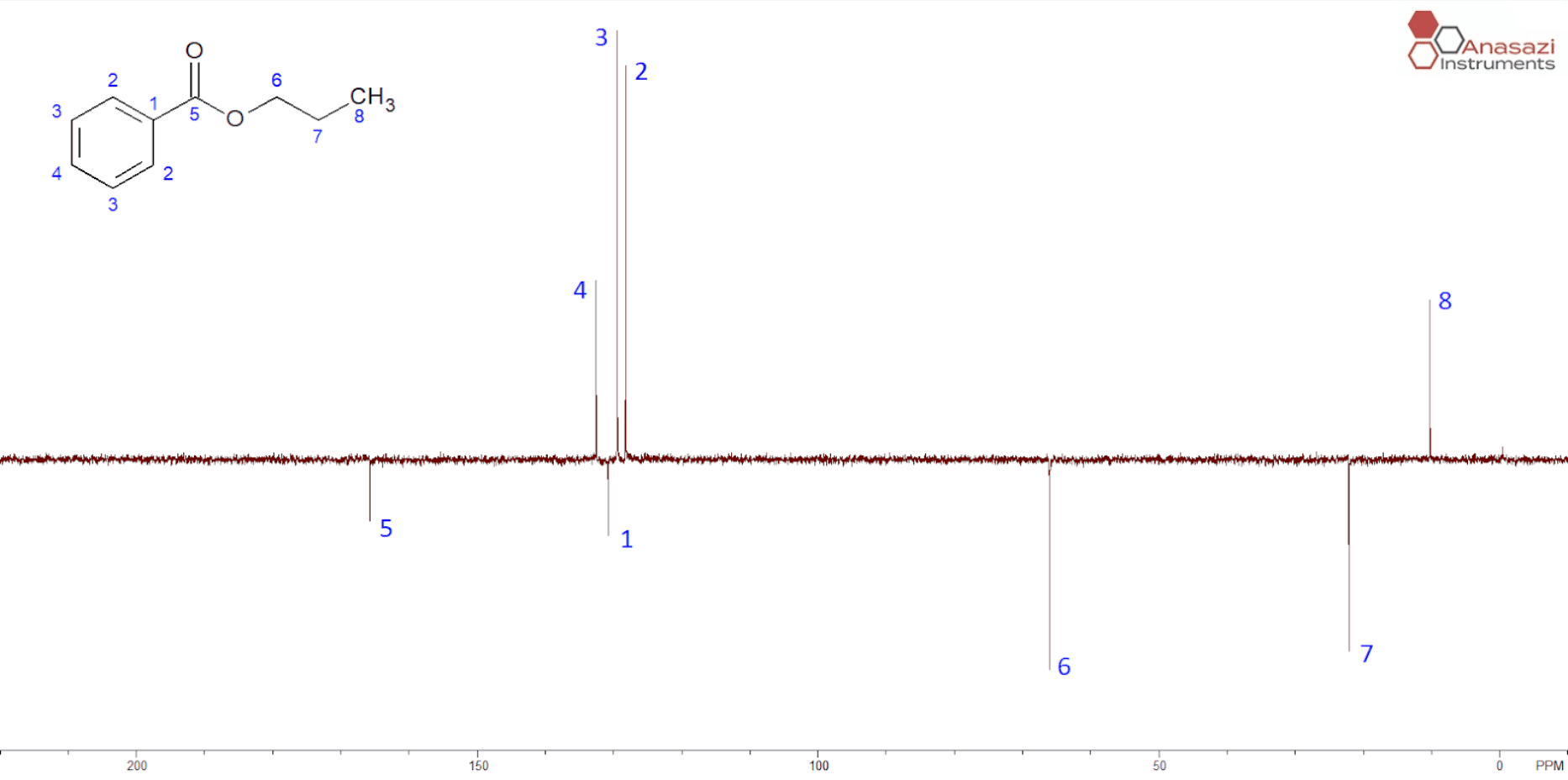
The main advantage of the APT is the inclusion of peaks for quaternary carbons (without directly attached protons). In a molecule like propylbenzoate (See Figure 2), the additional information would allow you to skip collecting the 13C data completely and just run the APT experiment. Additionally, the amount of time required to collect and process the data is the same as running a single 13C experiment. Because one gets chemical shift information for all the peaks plus the multiplicity of the attached protons, this can be a tremendous timesaver in the lab.
APT NMR Spectra Explained
Figure 2 shows the results from the standard APT sequence used on the EFT spectrometer. On modern NMR spectrometers, you can acquire and auto-process the spectrum with a single command (APT). Like the DEPT experiment, the peaks for CH (2, 3 and 4) and CH3 (8) point up while the peaks for the CH2 (6 and 7) point down. Additionally, the peaks for the carbons without directly attached protons (1 and 5) also point down.
Figure 2
APT NMR Disadvantages
The main disadvantage of the APT experiment is similar to that of acquiring carbon data. The T1 for protonated carbons is relatively short, whereas non-protonated carbons take 60-90 seconds to relax. Therefore, the peaks for carbons without directly attached protons (1 and 5) are always smaller.
The APT experiment does not distinguish between CH and CH3 peaks. If the compound structure was completely unknown, one might be fooled into thinking that a CH was a CH3 and vice versa.
DEPT or APT
Both DEPT and APT are useful methods for structural elucidation. DEPT-45, DEPT-90 and DEPT-135 together reveal primary, secondary and tertiary carbons but do not reveal quaternary carbons. APT reveals primary, secondary, tertiary and quaternary carbons but does not differentiate between methyl and methine carbons as does DEPT.
Which one is better depends on your research and what you need to know. Do you have any other questions about NMR?
Feel free to contact us.
